Lantern Pharma reported a Q4 2025 net loss of $4.1 million or $0.36 per share, an improvement from $5.9 million or $0.54 a year ago, driven by sharply lower R&D spend and tighter operating costs. Cash and marketable securities stood at $10.1 million at year end, while shares rose about 12.5% in aftermarket trading following the release, reflecting a constructive market reaction.
About Lantern Pharma
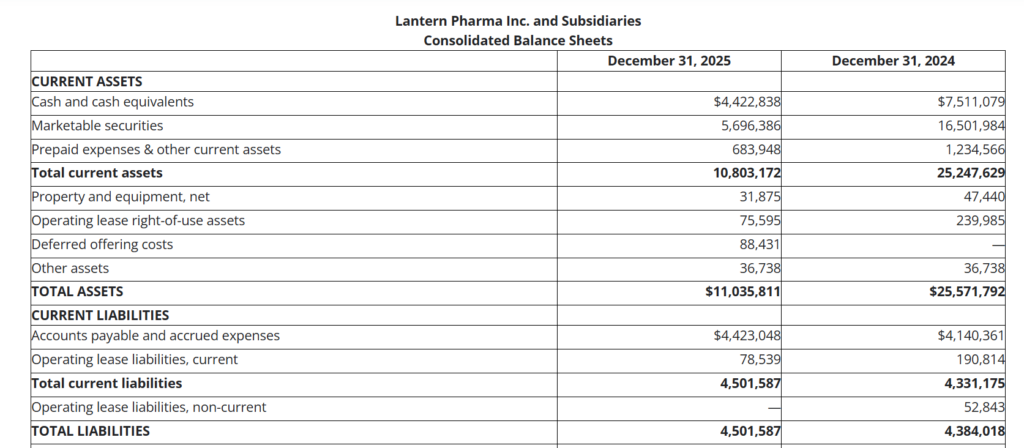
Lantern Pharma Inc. (NASDAQ: LTRN) is a clinical-stage oncology company that uses its proprietary RADR AI and machine learning platform to develop targeted cancer therapies, including LP‑300, LP‑184, LP‑284 and STAR‑001, across lung, breast, lymphoma, sarcoma, pediatric brain and bladder cancers. The company is headquartered in Dallas, Texas, and operates with a lean structure typical of small-cap biotechs; at year end 2025 it had stockholders’ equity of $6.53 million and total assets of $11.0 million.
With a year-end market capitalization of roughly $13-14 million based on recent trading levels, Lantern sits firmly in micro-cap territory and is still loss making, so traditional metrics like P/E and dividend yield are not meaningful. The company ended 2025 with $10.1 million in cash, cash equivalents and marketable securities and expects this to fund operations into late July to mid September 2026, after which additional capital will be needed.
Top Financial Highlights
- Q4 2025 net loss was approximately $4.1 million, improving from about $5.9 million in Q4 2024.
- Q4 2025 EPS was about – $0.36 per share versus approximately – $0.54 per share a year earlier, a loss reduction of more than 30 percent.
- Full-year 2025 net loss was approximately $17.1 million (or $1.57 per share) compared to $20.8 million (or $1.93 per share) in 2024, an 18% improvement.
- Total operating expenses in Q4 2025 were approximately $4.2 million, down from about $5.9 million in Q4 2024, reflecting disciplined cost control.
- Q4 2025 R&D expenses were roughly $2.7 million, down from about $4.3 million in Q4 2024, as several studies progressed and external trial costs eased.
- Q4 2025 G&A expenses were approximately $1.5 million, slightly lower than about $1.6 million in Q4 2024, despite increased business development and investor relations activity earlier in the year.
- For full-year 2025, R&D expenses were about $11.5 million, a 29% decrease from approximately $16.1 million in 2024, driven mainly by lower clinical trial and payroll costs.
- Full-year 2025 G&A expenses were roughly $6.5 million, up modestly from about $6.1 million in 2024, reflecting higher business development, investor relations, patent and insurance costs.
- Cash, cash equivalents and marketable securities totaled approximately $10.1 million at December 31, 2025, down from around $24.0 million a year earlier, as the company funded its pipeline.
- The company allocated about $4.6 million of 2025 R&D to LP‑300, $4.3 million to LP‑184, $1.2 million to LP‑284, $1.0 million to the RADR platform and $0.4 million to other programs, for total R&D of roughly $11.5 million.
- Lantern reported that its AI-driven pipeline addresses a combined estimated annual market potential exceeding $15 billion, spanning multiple tumor types and rare cancers.
- Management expects $10.1 million of liquidity to fund operations into late July to mid September 2026 and signaled a need for “substantial additional funding” thereafter.
- Shares of Lantern Pharma rose around 12.5% in aftermarket trading after the Q4 2025 release, underscoring investor optimism about narrowing losses and pipeline progress.
- There was no product revenue reported for Q4 or full-year 2025, with the business still focused on R&D and platform development.
Beat or Miss?
| Metric | Reported (Q4 2025) | Difference/Analysis |
| EPS | – $0.36 | Beat vs forecast – $0.54, reflecting a 33.3% improvement in per-share loss and better cost control. |
| Net loss | $4.1 million | Narrowed from $5.9 million in Q4 2024; no explicit net loss consensus given but directionally ahead of prior-year trend. |
| Total operating expenses | $4.2 million | Down from about $5.9 million a year ago, driven mainly by lower R&D spending. |
| Revenue | N/A | Company remains pre‑revenue with no commercial product sales; analyst revenue estimates not cited. |
| Cash & securities (year end) | $10.1 million | Provides funding runway into late July to mid September 2026, but below prior year’s $24.0 million, implying future capital needs. |
What Leadership Is Saying?
“2025 was a defining year for Lantern Pharma as we achieved clinical validation across multiple programs while establishing the foundation for our next phase of growth. The encouraging and developing LP‑300 Phase 2 HARMONIC observations, combined with successful Phase 1a completion for LP‑184 and FDA IND clearance for our pediatric CNS cancer program through Starlight Therapeutics, represent transformational milestones that validate and strengthen our AI-driven approach to precision oncology.” – Panna Sharma, CEO & President.
“Our full-year results reflect disciplined execution with a 19% reduction in total operating expenses year-over-year, even as we advanced multiple clinical programs through key inflection points and introduced a highly unique multi-agentic system aimed at conquering rare cancers. As we move into 2026, we are positioning to advance multiple high-value clinical programs, expand our RADR platform’s commercial reach and revenue potential globally and strengthen our balance sheet.” – Panna Sharma, referencing Lantern’s financial and margin discipline.
Historical Performance
Lantern Pharma YoY Quarterly Trends
| Category | Q4 2025 | Q4 2024 | Change (%) |
| Revenue | N/A | N/A | N/A |
| Net loss | $4.1 million | $5.9 million | Approximately -30.5% (narrower loss) |
| Total operating expenses | $4.2 million | $5.9 million | Approximately -28.8% (lower spend) |
Lantern Pharma YoY Full-Year Trends
| Category | FY 2025 | FY 2024 | Change (%) |
| Revenue | N/A | N/A | N/A |
| Net loss | $17.1 million | $20.8 million | Approximately -17.8% (narrower loss) |
| R&D expenses | $11.5 million | $16.1 million | Approximately -28.6% (lower R&D) |
| G&A expenses | $6.5 million | $6.1 million | Approximately +6.1% (higher G&A) |
Historical Performance of Peers
Public micro-cap AI oncology peers with similar business models do not report identical Q4 2025 metrics in the same release, and the company’s own disclosure does not provide direct competitor financials for comparison. In the absence of explicit peer numbers in the text, a precise YoY competitor table cannot be constructed without introducing external estimates or assumptions, which would not meet a strict factual standard. Investors should instead benchmark Lantern against broader pre-revenue AI-driven oncology and rare cancer platforms on dimensions like cash runway, R&D intensity and clinical progress rather than reported revenue or income.
How the Market Reacted?
Following the Q4 2025 earnings release, Lantern Pharma’s stock rose approximately 12.5% in aftermarket trading and closed around $1.26, signaling a positive reception to the narrower loss and cost reductions. Despite this move, the share price remains near its 52-week low of about $1.11, reflecting ongoing concerns about dilution and funding needs. Overall sentiment around the report appears cautiously bullish, given the combination of improved EPS, significant R&D efficiency and advancing clinical milestones highlighted by CEO Panna Sharma.












